Thus, as size (atomic radius) increases, the ionization energy should decrease. For larger atoms, the most loosely bound electron is located farthest from the nucleus and so is easier to remove. Moving down a group, atoms add electron shells so the atomic radius increases and it takes less energy to remove electrons. Metallic character increases moving down a periodic table group and decreases moving across a period. Periodic trends in atomic size, ionization energy, and electronegativity are described. Energy is always required to remove electrons from atoms or ions, so ionization processes are endothermic and IE values are always positive. Both metallic and nonmetallic character are periodic table trends. This page titled 4.1.2: The periodic trends in properties of the elements is shared under a Public Domain license and was authored, remixed, and/or curated by Muhammad Arif Malik. Consequently, all transition-metal cations possess d n valence electron configurations, as shown in Table 23.2 for the 2+ ions of the first-row transition metals. This would be an element in group 6, such as oxygen. It is likely to react with an element with six valence electrons that wants to gain two electrons. Therefore, it is very reactive and gives up electrons in chemical reactions. The energy required to remove the third electron is the third ionization energy, and so on. And because elements with similar valence electrons will have similar reactivities, they will form similar ions. The loss of one or more electrons reverses the relative energies of the ns and (n 1)d subshells, making the latter lower in energy. A: Calcium is a group 2 element with two valence electrons. The highest electronegativity value is for fluorine. Electronegativities generally decrease from the top to bottom of a group. 1, reacts readily with metals to form compounds that can be broadly classified as salts and, therefore, are known as the halogens, which is derived from a combination of Greek words that translate to 'salt makers. Electronegativity values generally increase from left to right across the periodic table. Since metals have few valence electrons, they tend to increase their stability by losing electrons to become cations. Each of the elements found in this column, which are boxed in lavender in Figure 2.2.1 2.2. 
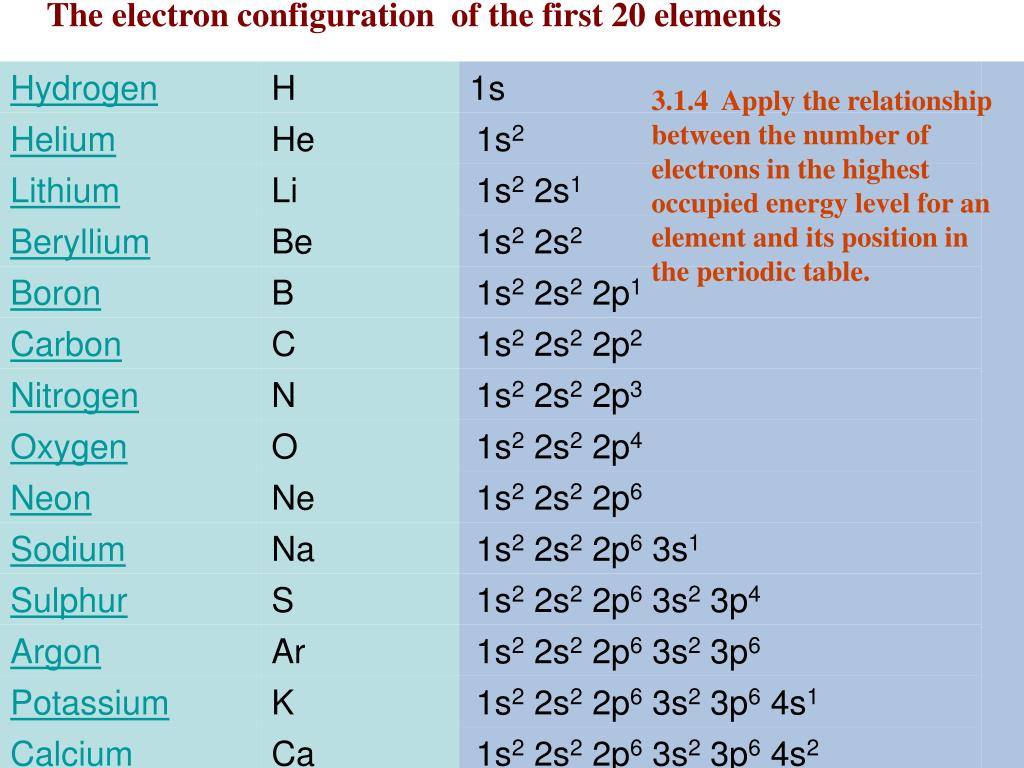
The number of valence electrons increases from top to bottom within each group. 
Atomic radius is the distance from the atom’s nucleus to the outer edge of the electron cloud. Electronegativity is a measure of the ability of an atom to attract the electrons when the atom is part of a compound. The number of valence electrons decreases from left to right across each period. Valence electrons are the electrons in the outermost shell, or energy level, of an atom. As Z increases by a small interval, the shell number increases, and so the electrons in the valence shell are much farther from the nucleus and are more shielded by the electrons in the lower energy shells.\] Electron-electron repulsions: due to their like charges, electron pairs orient themselves as far away as possible from each other, causing the electron cloud to expand (justifies trends across a period). \), we can see that when Z increases by one proton and the period increases (for example, going from Ne to Na), there is a relatively large decrease in Z eff. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
